U.S. Retina Surgeons Can't Stop Singing The Blues
AT A GLANCE
- Brilliant blue G is a vital dye that was recently approved by the US FDA under the name TissueBlue (Dutch Ophthalmic USA).
- US retina surgeons may now access an on-label vital dye for ILM staining.
- Among the conveniences of TissueBlue are its prefilled syringe, sharp contrast on the ILM, and elimination of the need to inject the dye under air or with infusion turned off.
Vital dyes are an essential tool of retina surgeons who perform macular surgery, specifically peeling of epiretinal membranes (ERM) or the internal limiting membrane (ILM). According to Market Scope, ILM peeling is performed in about a quarter of all vitrectomies.
Goals for membrane staining include reliable and adequate staining, good contrast with surrounding tissue, easy application, and safety of underlying retinal tissue.
In 2011 and 2015, I reviewed membrane staining options available in the United States at the time, including indocyanine green (ICG) (IC-Green, Akorn), triamcinolone, and compounded brilliant blue G (BBG).2,3 Although a commercially available brilliant blue product known as ILMBlue (DORC) has been an option for surgeons outside of the United States, not until recently has a US FDA–approved product been available in the United States.
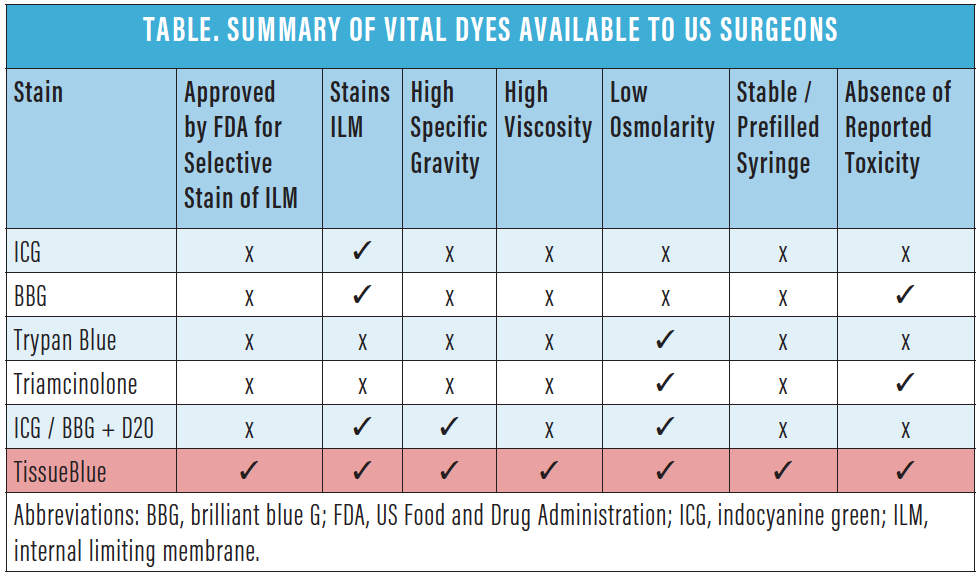
Let’s briefly summarize these vital dyes (Table).
ICG
ICG is used off-label for ILM staining. Contrast is dependent on ICG concentration, which involves a trade-off between safety and efficacy: A more concentrated solution, while providing more robust staining, risks toxicity to the exposed RPE during macular hole surgery.4
TRIAMCINOLONE
Triamcinolone, also used off-label, isn’t an actual stain. Microparticles settling on the membrane surface provide a demarcation of peeled and unpeeled membrane.
COMPOUNDED BBG
Compounded BBG, also off-label in the United States, must be supplied via a compounding pharmacy and may vary in preparation and safety. It has been reported that under limited FDA guidance, compounding pharmacies, if not vigilant, may provide contaminated product.5
TissueBlue
In December 2019, the FDA approved TissueBlue (0.025% brilliant blue G, Dutch Ophthalmic USA) for selective ILM staining. This approval finally provides access to a product that colleagues in Europe and globally have used for more than 350,000 cases since its launch in 2010.
As an FDA-approved product, TissueBlue meets active pharmaceutical ingredient standards, assuring levels of purity and consistency in manufacture. Outside of the United States, TissueBlue is marketed under the brand ILMBlue, which has a proven track record for safety and high-performance staining. The patented formulation of TissueBlue is unique in containing 4% polyethylene glycol (PEG). PEG is the carrier that is used to ensure that the staining agent maintains a tight, cohesive ball of dye as it falls to the back of the eye, due to its higher specific gravity compared with balanced salt solution, thereby avoiding the need for a fluid-air exchange during surgery.
WHAT I’VE LEARNED
In my experience, the density of TissueBlue provides a pooling effect on the posterior pole with robust ILM visualization 30 to 40 seconds after initial infusion. The PEG is critical to provide both higher density and higher specific gravity so that the dye settles through gravity to the back of the eye and also spreads in a flat wave across the macula without dispersing in the vitreous cavity.
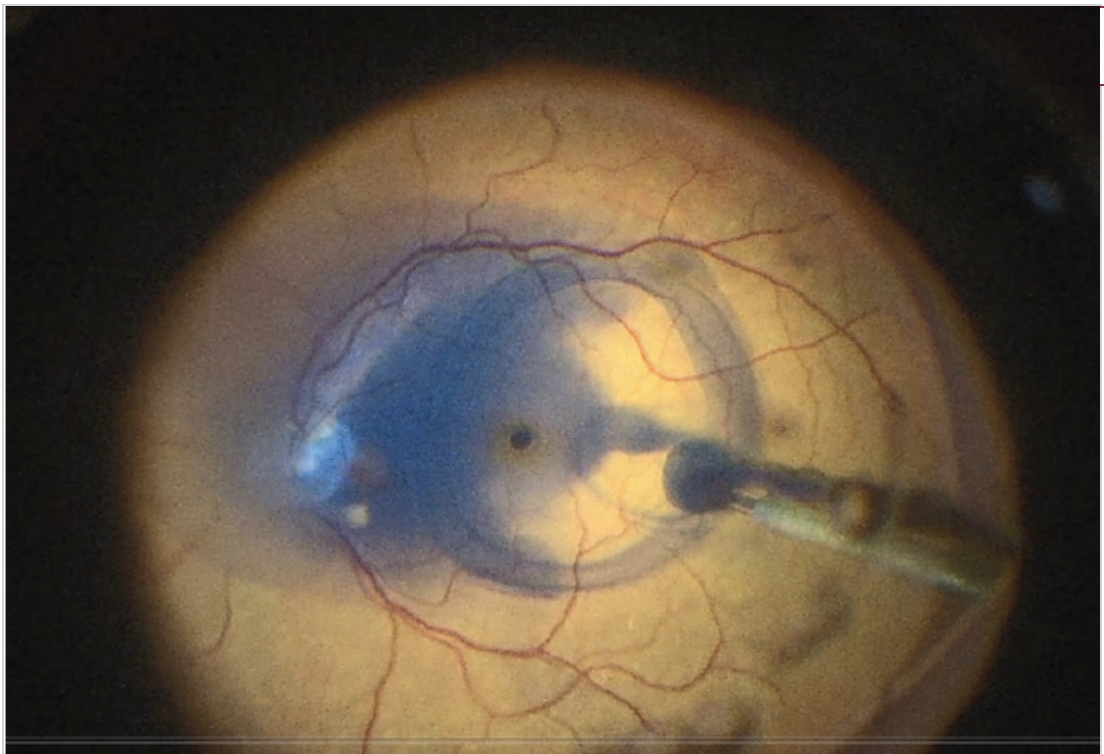
Because TissueBlue settles over the posterior pole, it is not necessary to turn off infusion to the eye or inject the dye under air—measures often needed to keep a dye over the macula and avoid dispersal after injection (Figure 1).
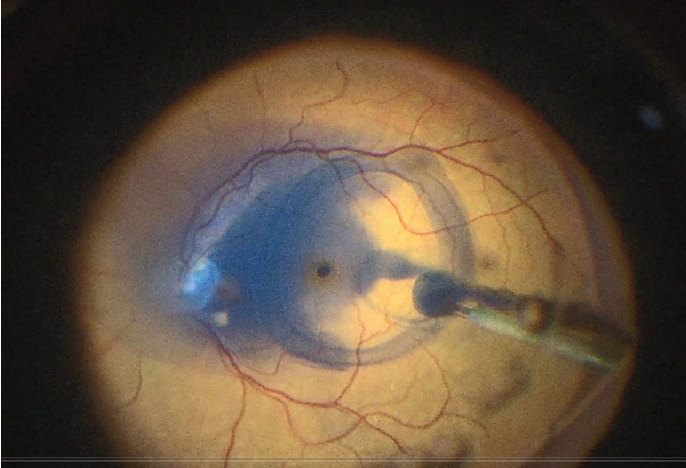
Figure 1. Pooling of dye over the posterior pole.
The dark blue color of the stained ILM stands in sharp contrast to the underlying retina when peeling commences (Figure 2). Additionally, because the stain is selective to the ILM, any areas covered by ERM are clearly visible via negative staining in contrast to the stained areas of ILM. With its proven safety profile in more than 300,000 cases, I have no concerns regarding restaining to confirm complete removal of the ILM if there is any doubt about the completeness of the peel. Finally, for the surgical staff, the availability of TissueBlue in a prefilled syringe is a major advantage: It is convenient and obviates the need to prepare or mix the dye prior to use (Figure 3).
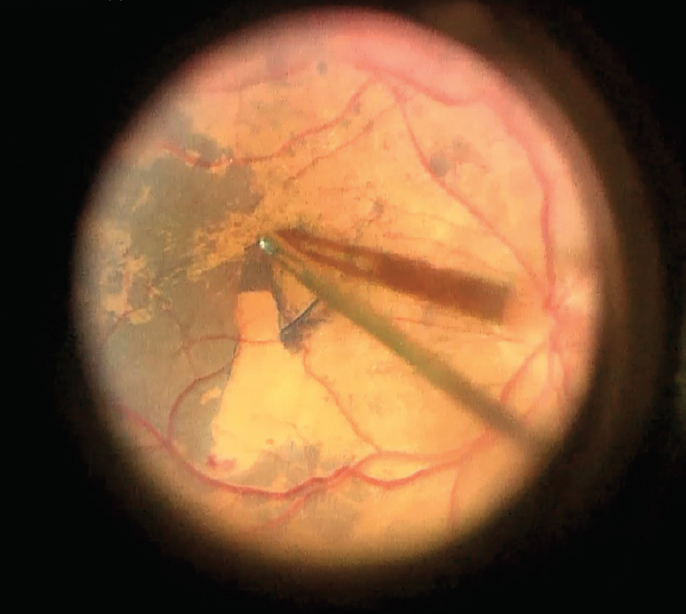
Figure 2. Contrast between stained ILM and underlying retina.
Figure 3. TissueBlue in a prefilled syringe.
For many years, I have advocated for the use of BBG in vitreoretinal surgery for membrane staining, so the availability of a version that is approved by the FDA for staining the ILM and in a formulation that is designed specifically to meet the needs of vitreoretinal surgeons is a significant advance. I have no reservations in recommending TissueBlue for ILM staining. This will be a useful new tool for vitreoretinal surgeons in the United States, who no longer must sing the blues over the lack of brilliant blue in their ORs.
AUTHOR: Brian C. Joondeph, MD, MPS
Partner, Colorado Retina Associates, Denver
[email protected]
Financial disclosure: None
REFERENCES:'
- Market Scope. 2018 Retinal Surgical Device Report. Table 21. Available at: www.market-scope.com.
- Joondeph BC. Vitreoretinal staining solutions (part 1 of 3). Retina Today. 2015;10(6):40-41.
- Joondeph BC. Uses of visualization aids in vitreoretinal surgery. Retina Today. 2011;6(6)70-72.
- Stanescu-Segall D, Jackson TL. Vital staining with ICG: a review of the clinical and experimental studies relating to safety. Eye (Lond). 2009;23(3):504-518.
- Mikosz CA, Smith RM, Kim M, et al. Fungal endophthalmitis associated with compounded products. Emerg Infect Dis. 2014;20(2):248-256.
CREDIT: July/August 2020 | Surgical Pearls in Retina Today

