C5 Inhibitor Avacincaptad Pegol For Geographic Atrophy Due To Age-related Macular Degeneration: A Randomized Pivotal Phase 2/3 Trial
Highlights
- Avacincaptad pegol, a complement C5 inhibitor, met its prespecified primary efficacy endpoint in reducing the mean GA growth rate in participants with AMD and was well tolerated in a randomized phase 2/3 pivotal trial.
Abstract
Purpose
The complement pathway may play a key role in the pathogenesis of age-related macular degeneration (AMD). The safety and efficacy of avacincaptad pegol (Zimura®), a C5 inhibitor, was assessed in participants with geographic atrophy (GA) secondary to AMD (GATHER1 Study).
Design
International, prospective, randomized, double-masked, sham-controlled, pivotal phase 2/3 clinical trial.
Participants
286 participants with GA secondary to AMD.
Main Outcome Measures
The primary efficacy endpoint was the mean rate of change in GA over 12 months measured by fundus autofluorescence (FAF) at three time points: baseline, month 6, and month 12.
Results
The reduction in the mean rate of GA growth (square root transformation) over 12 months was 27.4% (p-value = 0.0072) for the avacincaptad pegol 2 mg cohort and 27.8% (p-value = 0.005) for the avacincaptad pegol 4 mg cohort as compared to their corresponding sham cohorts. The results for both dose groups were statistically significant. Avacincaptad pegol was generally well tolerated after monthly administration over 12 months. There were no avacincaptad pegol related adverse events or inflammation. Further, there were no ocular serious adverse events and no cases of endophthalmitis. The most frequent ocular adverse events were related to the injection procedure.
Conclusions
Intravitreal administration of avacincaptad pegol 2 mg and 4 mg led to a significant reduction of GA growth in eyes with AMD over a 12 month period. As C5 inhibition theoretically preserves C3 activity it may offer additional safety advantages. A second confirmatory pivotal clinical trial is underway to confirm the efficacy and safety of avacincaptad pegol in slowing the GA growth (GATHER2 Study).
ARTICLE:
Age-related macular degeneration (AMD) is the leading cause of visual loss in individuals more than 50 years of age in developed countries.In the United States, it is estimated that approximately 11 million individuals are affected by AMD with a global prevalence of 170 million individuals. Because of the increased life expectancy in developed and developing countries, the elderly sector of the general population is expected to increase at the greatest rate in the coming decades. While 1 in 8 Americans was considered to be elderly in 1994, it is expected that 1 in 5 will fall into this category by 2030.Projections based on U.S. Census Bureau data suggest that the number of Americans over the age of 65 will more than double, increasing to 80 million by the middle of this century.
With increased age, AMD may progress to an advanced form, geographic atrophy (GA), a significant cause of bilateral, irreversible and severe loss of vision. On average, eyes with GA may lose 22 Early Treatment Diabetic Retinopathy Study (ETDRS) visual acuity (VA) chart letters over 5 years significantly reducing functional vision, quality of life, and independence.The median time to develop central GA in the first eye from the time of diagnosis is 2.5 years and the median time to develop central GA in the fellow eye is approximately 7 years.
Furthermore, development or progression of GA over time is a common cause of visual acuity loss in eyes diagnosed with neovascular (wet) AMD and treated with anti-vascular endothelial growth factor (VEGF) therapy. These data indicate that in many patients, regardless of whether they have the non-neovascular (dry) or the neovascular (wet) form of AMD, geographic atrophy may be the final anatomic outcome leading to loss of vision.
In GA, degeneration of macular photoreceptors, retinal pigment epithelial (RPE) cells and choriocapillaris results in marked thinning and/or atrophy of retinal tissue, leading to irreversible severe vision loss.
The complement cascade, which was originally described over a century ago by Paul Ehrlich, represents a critical portion of the innate immune system, involving a complex system of over 50 serum proteins.In addition to its role in the innate immune system, recent studies indicate that complement may also play a role in the adaptive immune system, cell and tissue development, homeostasis and repair.
The complement cascade is activated via the classical (antibody-dependent), the alternative (antibody-independent), and the lectin pathways.The three cascades converge to generate complement C3 convertase which cleaves complement C3 and leads to subsequent generation of complement C5 convertase which cleaves complement C5 into C5a and C5b, the key terminal effector components of the complement cascade, leading to cell death. Complement is thought to play a key role in retinal degeneration secondary to AMD based on genetic linkage studies, pre-clinical in vitro studies, and immunohistochemical studies that show that complement is deposited in post-mortem human eyes of patients diagnosed with AMD. Genetic linkage and association studies suggest that approximately 50% of eyes with dry AMD have polymorphisms in complement regulatory proteins compared to age-matched controls. Furthermore, polymorphisms in genes coding for complement or complement regulatory proteins confer an increased risk for AMD. In individuals homozygous for the risk allele, the likelihood of AMD is increased by a factor of 7.4.
Avacincaptad pegol, a pegylated RNA aptamer, is a potent and specific inhibitor of complement C5. Avacincaptad pegol inhibits cleavage of C5, a central component of the complement cascade, that plays multiple roles in innate immunity and cell death. Inhibition of this critical step in the complement cascade prevents the formation of key terminal fragments (C5a and C5b) regardless of the initial activation pathway (alternate, classical, or lectin). The C5a fragment may play an important role in inflammasome priming and activation leading to pyroptosis and cell death.C5b may be involved in the formation of membrane attack complex (MAC: C5b-9) which also leads to cell lysis and cell death. By inhibiting these C5-mediated activities, avacincaptad pegol may have the potential to slow down the progression of retinal cell degeneration and achieve therapeutic benefit in GA secondary to AMD.
Although anti-VEGF therapy is available for treatment of wet AMD, currently no Food and Drug Administration (FDA) or European Medicines Agency (EMA) approved treatment is available for GA. Herein, the 12-month results of a phase 2/3 pivotal trial, assessing the safety and efficacy of intravitreal administration of avacincaptad pegol in slowing the GA growth in association with AMD are presented. Based on the results of this trial, a confirmatory phase 3 trial is underway comparing avacincaptad pegol 2 mg with a sham-control group (GATHER2 Study).
Methods
Study Design
This international, prospective, randomized, double-masked, sham-controlled phase 2/3 pivotal clinical trial enrolled participants with GA secondary to AMD at 63 sites (US, Europe, and Israel) between January 2016 and October 2018. A list of study sites and investigators is available in the Appendix. The study was performed in accordance with the tenets of the Declaration of Helsinki, and International Conference on Harmonization Good Clinical Practice guidelines. The appropriate ethics committee or institutional review board at each study center approved the protocol. Informed consent was obtained from all participants. All data were collected in a Health Insurance Portability and Accountability Act-compliant manner. An Independent Data Monitoring Committee reviewed participant safety data during the course of the trial.
The trial consisted of two parts: in Part 1, patients were randomized in a 1:1:1 ratio to the following dose groups: avacincaptad pegol 1 mg, avacincaptad pegol 2 mg, and sham. Subsequently in Part 2, patients were randomized in a 1:2:2 ratio to the following dose groups: avacincaptad pegol 2 mg, avacincaptad pegol 4 mg, and sham. Due to the drug viscocity, avacincaptad pegol 4 mg dose was administered as two separate intravitreal injections of avacincaptad pegol 2 mg in 100 microliter volume each. To minimize bias in Part 2, participants who were in the avacinacptad pegol 2 mg arm also received an additional sham administration (to mimic two injections) and participants who were in the sham arm, received two sham administrations.
A web-based randomization system (ID-net™, IDDI, Belgium) was used and a minimization method was applied to the randomization stratified by site, size of baseline GA, and pattern of fundus autofluorescence (FAF) at the junctional zone. The minimization method maintains the balance between all treatment groups overall and for each stratification criterion. Patients received monthly intravitreal injections of avacincaptad pegol and/or sham for a duration of 18 months. Patients, evaluating physicians, independent reading center personnel (Duke Reading Center, Duke University, Durham, NC), and sponsor personnel were masked to the treatment (sham or avacincaptad pegol) that each individual patient was receiving during the study. The masking was preserved throughout the duration of the clinical trial including both parts 1 and 2.
Patients
To be eligible, participants had to be at least 50 years of age of either gender, with best corrected visual acuity between 20/25 – 20/320 in the study eye. The GA had to be secondary to AMD, non-foveal-centered and in part within 1500 microns from the foveal center. The total GA area had to be ≥ 2.5 and ≤ 17.5 mm2 (1 and 7 disk areas [DA]), determined by screening FAF images, and had to be photographed in its entirety within field 2 (30 degree photographic field centered on the foveal center). For multifocal GA lesions, at least one focal lesion had to be ≥ 1.25 mm2 (0.5 DA).
Key ocular exclusion criteria included GA secondary to any condition other than AMD in either eye (e.g., drug-induced), any prior treatment for AMD (dry or wet) except oral supplements of vitamins and minerals, or any prior intravitreal treatment for any indication in either eye. Further exclusion criteria included evidence of choroidal neovascular membrane (CNV) in either eye, any ocular condition that would progress during the course of the study that could affect central vision or otherwise be a confounding factor, and any sign of diabetic retinopathy in either eye. A comprehensive list of inclusion and exclusion criteria is available in the Appendix.
Image Acquisition
Blue light fundus autofluorescence (FAF), fundus photographs (FPs), and fluorescein angiograms (FAs) were acquired with the modified 3-field imaging protocol (field 1M: 30 degree field centered on the temporal aspect of the optic nerve, field 2: 30 degree field centered on the foveal center, and field 3M: 30 degree field centered 1-1.5 disc diameter temporal to the center of field 2). A Heidelberg Spectralis or HRA (Heidelberg Engineering, Heidelberg, Germany) system was used to obtain FAF and near infrared field 2 images. Spectral domain optical coherence tomography (OCT) scans were obtained with Cirrus (Carl Zeiss Meditec, Jena, Germany) or Heidelberg Spectralis systems. Cirrus OCT scans were acquired with 512 X 128 macular cube- and 5-line HD raster scan protocols. Spectralis scans were obtained with 97-line volume scan (20° X 20°, high resolution mode, ART = 9) and 73 line volume scan (20° X 15°, high resolution mode, ART = 9) protocols.
Geographic Atrophy Area Measurement
Masked Readers (Duke Reading Center) independently analyzed and graded the images. Readers used RegionFinder software (Heidelberg Engineering) to quantify GA area on FAF images. Optical coherence tomography and near infrared imaging were used to supplement the FAF GA area analysis to help define GA boundaries and GA location relative to the foveal center. Two experienced primary readers independently measured GA area at each timepoint. If there was more than a 10% discrepancy in the measurements, the results were arbitrated by the Reading Center Director (GJJ).
Study Endpoints and Statistical Analysis
The prespecified primary efficacy endpoint was the mean rate of change in GA area over 12 months measured by fundus autofluorescence at three time points: baseline, month 6, and month 12. Square root transformation was applied to mitigate the impact of baseline lesion size on GA growth over time as previously described.
Secondary endpoints included the mean change in best corrected visual acuity (ETDRS letters) from baseline to month 12 and the mean change in low luminance best corrected visual acuity (ETDRS letters) from baseline to month 12.
Safety endpoints included adverse events (AEs), serious adverse events (SAEs), vital signs (pulse, systolic and diastolic blood pressure), ophthalmic findings including intraocular pressure (IOP), ophthalmic examination, OCT and FA findings, FAF, electrocardiogram (ECG) and laboratory variables including hematology, renal function, hepatic function, electrolytes and urinalysis.
All efficacy analyses were conducted for the intention-to-treat (ITT) population. The ITT population consisted of all randomized subjects who received at least one dose of the assigned treatment, sham, avacincaptad pegol 1mg, avacincaptad pegol 2 mg, or avacincaptad pegol 4 mg.
Based on a prespecified statistical analysis plan (SAP), the analyses were based on comparisons of avacincaptad pegol 2 mg vs its corresponding sham, and avacincaptad pegol 4 mg vs its corresponding sham. The Hochberg procedure was used to control for multiplicity. Although the avacincaptad pegol 1 mg dose was not included in the prespecified statistical analysis, a descriptive analysis for treatment effect of avacincaptad pegol 1 mg dose vs its corresponding sham from part 1 was conducted in a supportive manner. Since avacincaptad pegol 2 mg and corresponding sham arm were present in both part 1 and 2 of the trial, eyes randomized in part 1 were combined with the eyes randomized in part 2, and the analysis included a regression factor by part (part 1 vs part 2). The comparison of avacincaptad pegol 4 mg vs sham was based only on eyes randomized in part 2 since these eyes were only enrolled in part 2 of the study.
The design of this phase 2/3 pivotal trial was based on screening methodology presented in Fleming and Richardson.37 The prespecified primary analysis for each of the two pairwise comparisons of active dose of avacincaptad pegol vs the sham was formally determined with a three-category decision guideline based on whether the estimated mean reduction in rate of square root of GA area growth over 12 months for a dose of avacincaptad pegol vs sham was less than 14%, was between 14% and 24.5%, or was at least 24.5%. If the estimated reduction in the rate of GA growth rate over 12 months for a dose of avacincaptad pegol vs sham was less than 14%, then this dose of avacincaptad pegol was considered to be not plausibly more efficacious than the sham and its utility in GA secondary to AMD should be reconsidered. If the estimated reduction in GA growth rate over 12 months for a dose of avacincaptad pegol vs sham was between 14% and 24.5%, then this dose of avacincaptad pegol was considered to be plausibly more efficacious than the sham and should be evaluated in subsequent phase 3 pivotal clinical trials. If the estimated reduction in the rate of GA growth over 12 months for a dose of avacincaptad pegol vs sham was at least 24.5%, then this dose of avacincaptad pegol would be statistically significantly more effective than the sham, with strength of evidence meeting the standard requirement of a 0.0125 one-sided false positive error rate (incorporating an adjustment for multiplicity arising from comparing each dose with the sham). The latter scenario in this trial, whereby the estimated reduction in the GA growth rate compared to sham is statistically significant, would indicate that this study could count as one of the two required pivotal phase 3 trials for the registration of avacincaptad pegol.
The power calculations for the above scenarios were based on the released results from a clinical trial of a complement C3 inhibitor, including the estimated precision of the estimated effects on rate of GA growth over 12 months, including the plausible effect size for avacincaptad pegol.
The primary efficacy analysis was based on a Mixed-Effects Model for Repeated Measures (MMRM) model to compare the treatment groups. MMRM analysis does not require data imputation and uses only the observed data. The treatment effects estimated with MMRM are valid under the assumption of missingness at random (MAR). To assess the robustness of the primary efficacy results, a number of prespecified sensitivity analyses were performed to assess the potential magnitude and direction of the impact of missing data. Four simple sensitivity analyses described by Miller et al. were performed39: (1) the observed means from the active arm and the sham arm were imputed to participants with missing data in the arm to which they were allocated to; (2) the observed means from the active arm and the sham arm were imputed to participants with missing data in the opposite arm to which they were allocated; (3) the average of observed means from the active arm and the sham arm were imputed to all missing data; and (4) the observed mean from the sham arm was imputed to all missing data. The treatment effects remained statistically significant in these four sensitivity analyses. Next, a “shift imputation” approach consisted of replacing missing values by their expected values using multiple imputation, plus a specified treatment-dependent shift until statistical significance was lost, was performed to confirm the robustness of the results to missing values. Finally, the “pattern-mixture-model imputation” approach consisted of replacing missing values by values imputed under pattern-mixture-model restrictions was performed to investigate the potential impact of missingness not at random.
All safety analyses were performed on the safety population. The safety population included all subjects who received at least one dose of study drug. This clinical trial is registered with ClinicalTrials.gov (identifier, NCT02686658).
Results
Patients and Baseline Characteristics
A total 286 participants with GA secondary to AMD were enrolled. 77 participants were enrolled in part 1: 26 participants were randomized in the 1 mg cohort, 25 participants in the 2 mg cohort, and 26 participants in the sham cohort (Figure 1). 209 participants were enrolled in part 2: 42 participants were randomized in the 2 mg cohort, 83 participants in the 4 mg cohort, and 84 participants in the sham cohort (Figure 1). Imaging data collected from 223/260 (86%) participants who had at least two gradable FAF measurements (baseline and month 6 or month 12) were included in the MMRM analysis for the primary efficacy endpoint (Table S1). The Reading Center Reader intraclass correlations were high: 0.99 at baseline, 0.99 at month 6, and 0.99 at month 12 (1.0 indicating perfect correlation between the two readers). The percentage of eyes that required arbitration of their FAF images at baseline was 19.8%, at month 6 was 15.6%, and at month 12 was 9.8%. Baseline characteristics were balanced between various study groups (Table 1). Most participants were female, Caucasian, and had a mean age of 78.2 years. Participants in the sham, avacincaptad pegol 2 mg, and avacincaptad pegol 4 mg cohorts received on average 10.8, 10.5, and 9.8 treatments respectively out of 12 possible treatments during the first 12 months of the trial.
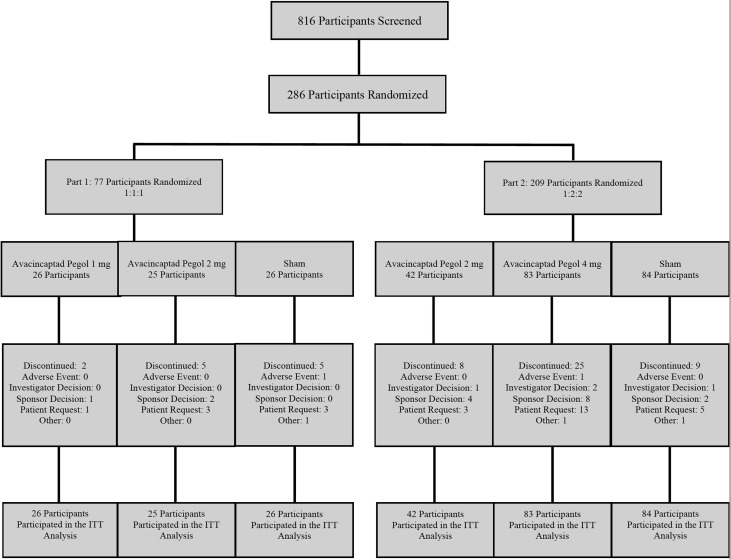
Figure 1 Clinical trial flowchart showing participant disposition. 286 participants were randomized in this study, which consisted of two parts: in Part 1, 77 participants were randomized in a 1:1:1 ratio to the following dose groups: 26 participants in avacincaptad pegol 1 mg, 25 participants in avacincaptad pegol 2 mg, and 26 participants in sham. In Part 2, 209 participants were randomized in a 1:2:2 ratio to the following dose groups: 42 participants in avacincaptad pegol 2 mg (2 mg and sham), 83 participants in avacincaptad pegol 4 mg (administered as two intravitreal injections of avacincaptad pegol 2 mg), and 84 participants in sham (sham and sham). Efficacy analysis was conducted for the intention-to-treat (ITT) patient population. Data from 86% of the participants were included in the primary efficacy endpoint analysis, participants who have had baseline and at least one additional gradable fundus autofluorescence measurement (at month 6 and/or month 12).
Table 1 Demographics and baseline characteristics (intention-to-treat [ITT] population).
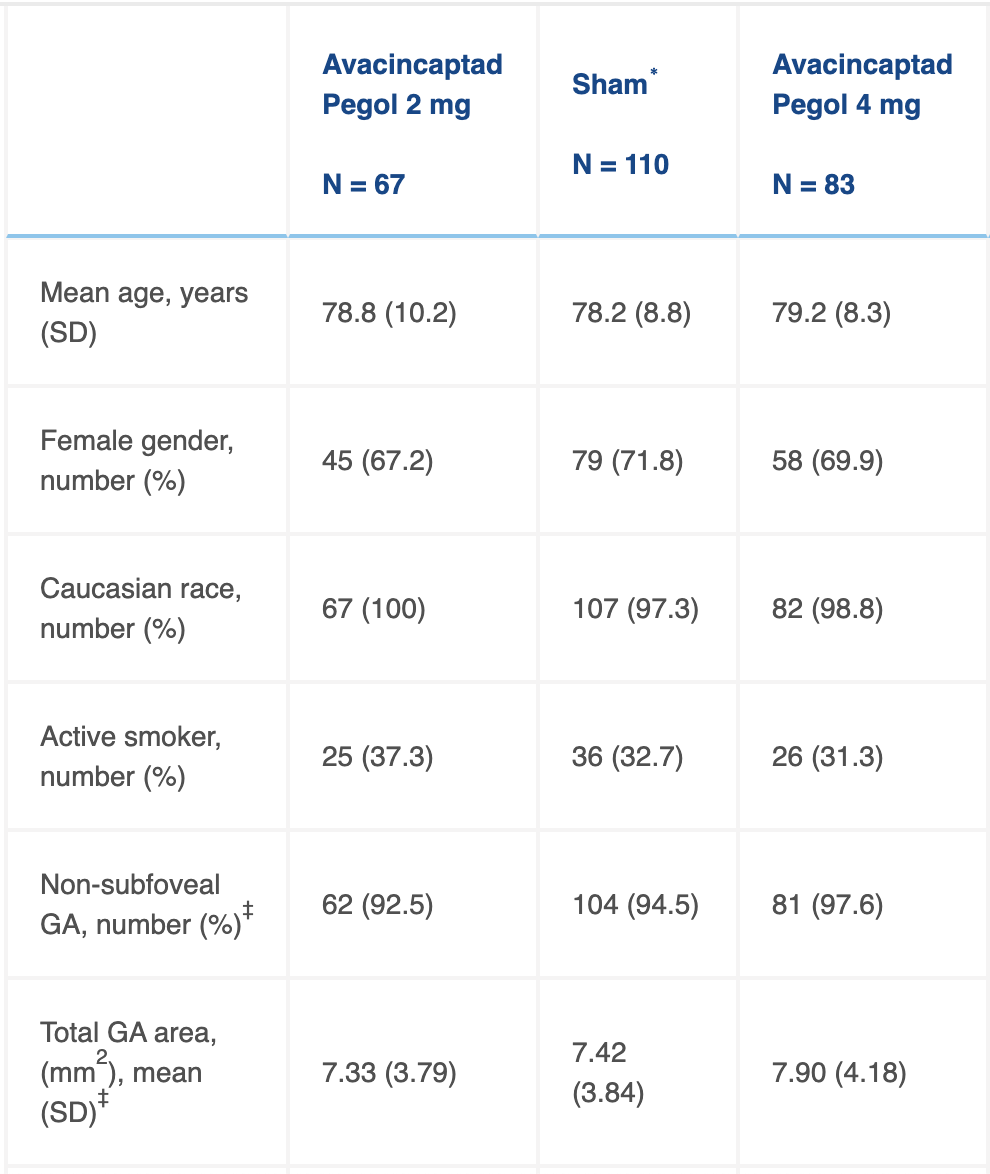
SD: standard deviation; ∗sham for 2 mg arm; ∗∗sham for 4 mg arm; ‡ Study eye; GA: geographic atrophy; BCVA: best-corrected visual acuity; ETDRS: Early Treatment Diabetic Retinopathy Study; LL-BCVA: low-luminance best-corrected visual acuity; LL-VD: low-luminance visual acuity deficit (BCVA minus LL-BCVA).
Efficacy Endpoint Analyses
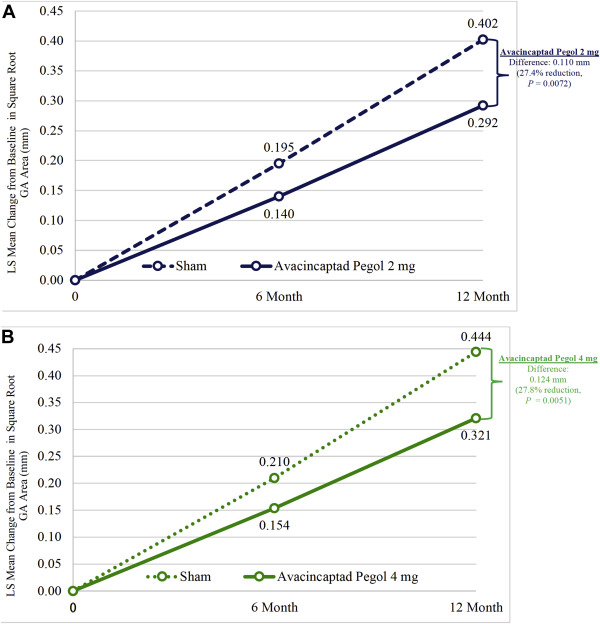
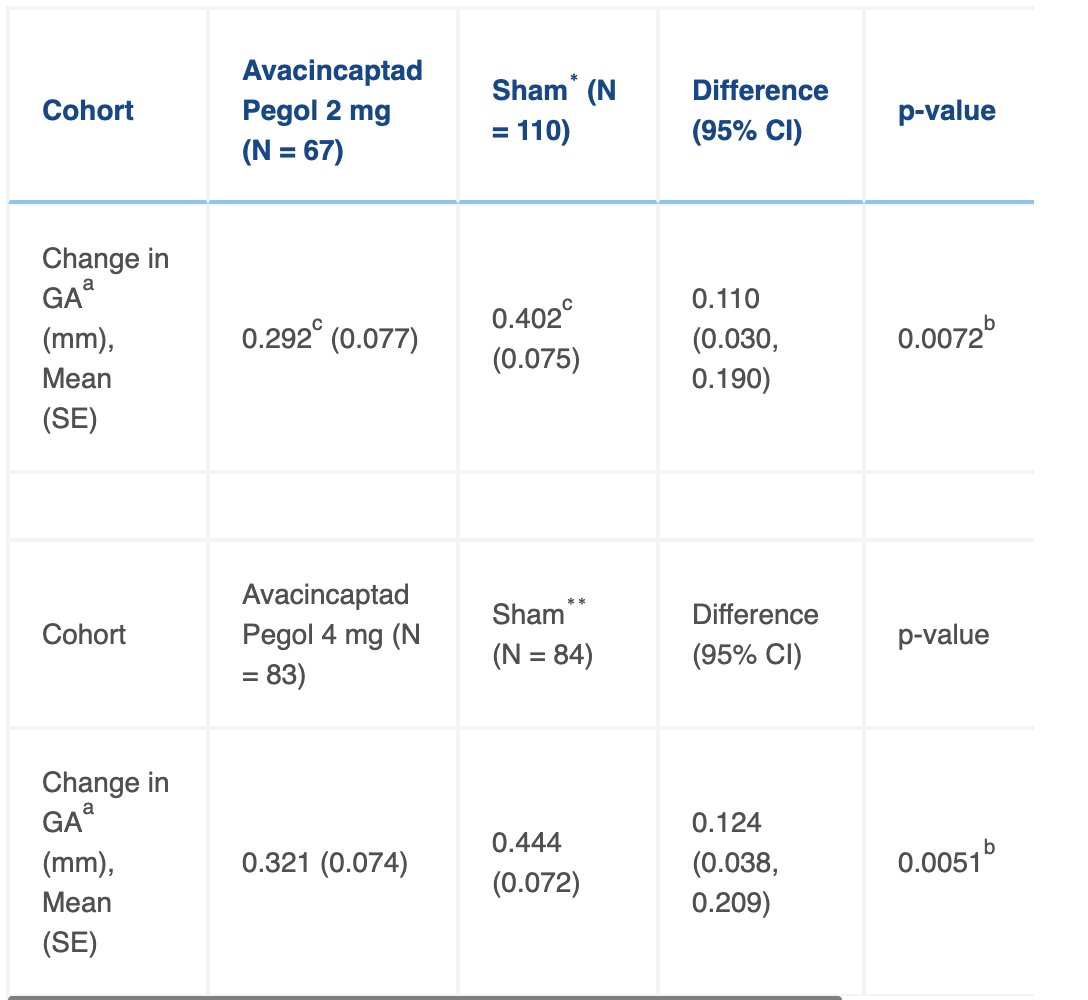
Both avacincaptad pegol 2 mg and avacincaptad pegol 4 mg met their prespecified primary efficacy endpoint, reducing the mean GA growth rate over 12 months. The primary efficacy endpoint was assessed using square root transformation of the GA lesion area. The least squares (LS) means change in the square root GA lesion area from baseline to month 12 for avacincaptad pegol 2 mg was 0.292 mm and for the corresponding sham was 0.402 mm and for avacincaptad pegol 4 mg was 0.321 mm and for the corresponding sham was 0.444 mm (Table 2). The reduction in the mean GA growth rate over 12 months was 27.4% (0.110 mm, p-value = 0.0072) for the avacincaptad pegol 2 mg group as compared to its corresponding sham-control group and 27.8% (0.124 mm, p-value = 0.0051) for the avacincaptad pegol 4 mg group as compared to its corresponding sham-control group, both meeting the prespecified significance level of p < 0.0125 one-sided, incorporating an adjustment for multiplicity arising from comparing each dose with their corresponding sham groups (Figures 2 A, B). The impact of avacincaptad pegol treatment on GA growth was evident as early as month 6 with a 28.4% reduction for avacincaptad 2 mg dose and a 26.6% reduction for avacincaptad pegol 4 mg when compared to their corresponding sham groups. Monthly treatment with avacincaptad pegol 2 mg and 4 mg also led to an observed untransformed (without square root transformation) 30.5% and 25.6% reduction in the mean GA growth rate respectively from baseline to month 12 when compared to their corresponding sham-control groups (Figures S1A and B, available at http://www.aaojournal.org).
Table 2Mean rate of change in geographic atrophy (GA) area from baseline to month 12 (square root transformation).
CohortAvacincaptad Pegol 2 mg (N = 67)Sham∗ (N = 110)Difference (95% CI)p-value% DifferenceChange in GAa (mm), Mean (SE)0.292c (0.077)0.402c (0.075)0.110 (0.030, 0.190)0.0072b27.4%CohortAvacincaptad Pegol 4 mg (N = 83)Sham∗∗ (N = 84)Difference (95% CI)p-value% DifferenceChange in GAa (mm), Mean (SE)0.321 (0.074)0.444 (0.072)0.124 (0.038, 0.209)0.0051b27.8%
SE: standard error; 95% CI: 95% confidence interval; ∗sham for 2 mg arm; ∗∗sham for 4 mg arm
(a) = Based on the least squares (LS) means from the mixed-effects repeated measures (MMRM) model.
(b) = Reflects statistically significant p-value; Hochberg procedure was used for significance testing.
(c) = The LS means is an estimate of the MMRM model, drawing on all available data, including data from groups with different randomization ratios in Part 1 and Part 2, and should not be interpreted as directly observed data.
Table 2 Mean rate of change in geographic atrophy (GA) area from baseline to month 12 (square root transformation).
SE: standard error; 95% CI: 95% confidence interval; ∗sham for 2 mg arm; ∗∗sham for 4 mg arm(a) = Based on the least squares (LS) means from the mixed-effects repeated measures (MMRM) model.(b) = Reflects statistically significant p-value; Hochberg procedure was used for significance testing.(c) = The LS means is an estimate of the MMRM model, drawing on all available data, including data from groups with different randomization ratios in Part 1 and Part 2, and should not be interpreted as directly observed data.
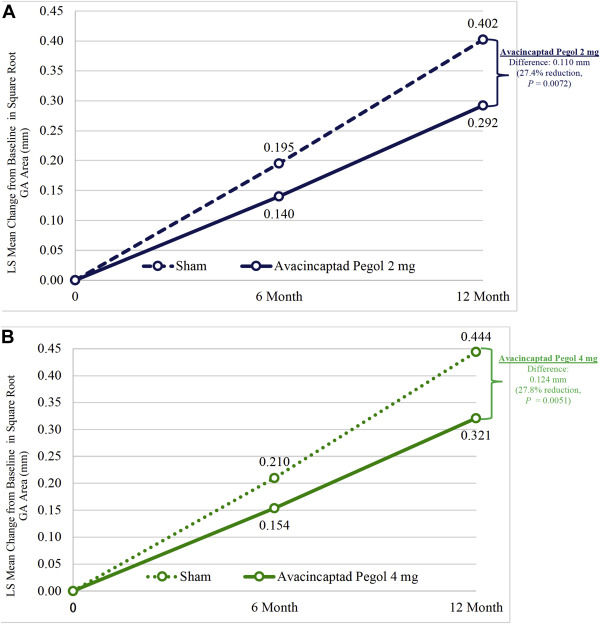
Figure 2A. The mean rate of change in square root of geographic atrophy (GA) area from baseline to month 12, measured at 3 timepoints with fundus autofluorescence. Least squares (LS) means based on estimates from a mixed-effects repeated measures (MMRM) model on available intention-to-treat (ITT) population. The p-value for avacincaptad pegol 2 mg vs the sham was 0.0072 and was statistically significant (27.4% reduction in GA growth). The 95% confidence interval for avacincaptad pegol 2 mg at month 6 was (0.053, 0.227) and at month 12 was (0.142, 0.442). The 95% confidence interval for the sham arm at month 6 was (0.109, 0.281) and at month 12 was (0.254, 0.549). Figure 2B. The mean rate of change in square root of geographic atrophy (GA) area from baseline to month 12, measured with fundus autofluorescence at 3 timepoints. Least squares (LS) means based on estimates from a mixed-effects repeated measures (MMRM) model on available intention-to-treat (ITT) population. The p-value for avacincaptad pegol 4 mg vs the sham was 0.0051 and was statistically significant (27.8% reduction in GA growth). The 95% confidence interval for avacincaptad pegol 4 mg at month 6 was (0.075, 0.233) and at month 12 was (0.175, 0.466). The 95% confidence interval for the sham arm at month 6 was (0.135, 0.286) and at month 12 was (0.304, 0.585).
Although efficacy data from participants receiving avacincaptad pegol 1 mg was not part of the prespecified statistical analysis, descriptive analysis indicated that, on average, the percent GA growth from baseline to month 12 for avacincaptad pegol 1 mg group was less than for the corresponding sham-control group (Table S2, available at http://www.aaojournal.org).
The detailed prespecified sensitivity analyses indicated that the results were robust and missing data had a small impact on the overall conclusions (Table S3, available at http://www.aaojournal.org). The shift imputation analyses indicated that statistical significance would only be lost after large shifts (a loss of > 40% of the observed treatment effect).
As expected from previous reports,avacincaptad pegol 2 mg or 4 mg did not have an impact on the mean change in best corrected visual acuity (BCVA) or low luminance best corrected visual acuity (LL-BCVA) from baseline to month 12, as compared to their corresponding sham groups (Tables 3 A and B). Of note, this trial was not designed to demonstrate a difference in mean change in BCVA or LL-BCVA at an overall one-sided statistical significance level of 0.025.
Table 3A. Mean change in best corrected visual acuity (Early Treatment Diabetic Retinopathy Study [ETDRS] letters) from baseline to month 12.
Safety Analyses
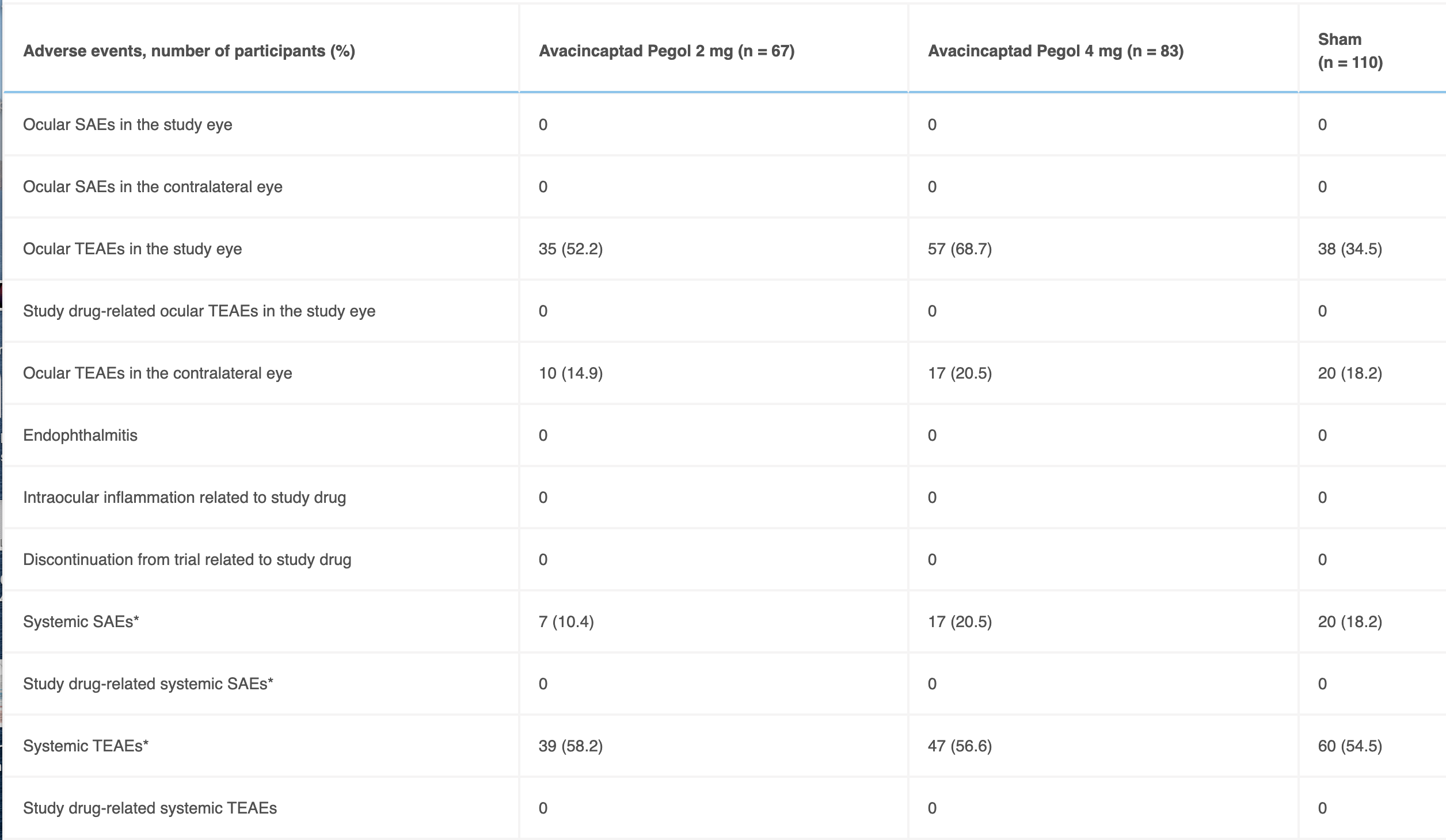
Avacincaptad pegol was generally well tolerated. There were no reported avacincaptad pegol-related adverse events or avacincaptad pegol-related inflammation (Table 4). There were no reported endophthalmitis or ocular serious adverse events (SAEs) in the study eye. The most frequently reported ocular adverse events (AEs) related to the injection procedure were conjunctival hemorrhage, conjunctival hyperemia, punctate keratitis, and increased intraocular pressure (IOP) (Table S4A, available at http://www.aaojournal.org). The mean pre-injection IOP was similar from baseline to month 12 across all arms. Transient elevation of mean IOP, consistent with the injection volume effect, was observed post-injection in participants receiving avacincaptad pegol, and mean IOP returned to near baseline levels at the next follow-up visit and at the end of the study. There were no reported AEs of glaucoma or ocular hypertension in subjects receiving avacincaptad pegol; one subject receiving sham had an AE of ocular hypertension.
Table 4Major reported safety data in the safety patient population. *Non-ocular
TEAE: treatment emergent adverse event, SAE: serious adverse event
Non-ocular adverse events were reported less frequently than ocular adverse events and were conditions that are known to commonly occur among the enrolled patient population, including urinary tract infection, fall, nasopharyngitis, and atrial fibrillation (Table S4B, available at http://www.aaojournal.org). Non-ocular SAEs were reported in 15.3% (27/176) of participants receiving avacincaptad pegol treatment, and 18.2% (20/110) of participants receiving sham treatment. None of the non-ocular treatment emergent adverse events (TEAEs) or SAEs were considered related to avacincaptad pegol treatment. There were no clinically meaningful vital sign changes, laboratory abnormality trends, or ECG changes.
Choroidal neovascular membrane (CNV) was reported by the investigators in the fellow eyes of 10 (3.5%) participants, in the study eye of 3 (2.7%) participants in the sham cohort, 1 (4%) patient in the avacincaptad pegol 1 mg cohort, 6 (9.0%) participants in the avacincaptad pegol 2 mg cohort, and 8 (9.6%) participants in the avacincaptad pegol 4 mg cohort. At the time of the design of the trial, it was thought that the development of CNV would hinder accurate FAF measurement analysis, and therefore participants who developed CNV during the trial were discontinued from the study.
Discussion
In this study, avacincaptad pegol, a complement C5 inhibitor, met its prespecified primary efficacy endpoint in reducing the mean GA growth rate in participants with AMD. The reduction in the mean GA growth rate over 12 months was 27.4% for the avacincaptad pegol 2 mg and 27.8% for the avacincaptad pegol 4 mg as compared to their corresponding sham-control groups. These results, together with a previous report,38 further validate the role of complement in the progression of AMD-associated GA and suggest a potential role for complement inhibitors to decrease GA growth rate. The GA growth rate in the sham arm of this trial was consistent with what has been reported in clinical trials for extrafoveal GA growth in dry AMD. As has been reported, extrafoveal location of GA is associated with faster growth rates compared with GA involving the fovea. The treatment effect was observed as early as month 6 and was maintained at month 12. The earlier onset of treatment effect, when compared to the literature for C3 inhibition, may be due to the further downstream inhibition of complement cascade at the C5 level.
Avacincaptad pegol was generally well tolerated after 12 months of administration with no avacincaptad pegol related adverse events or serious adverse events. Complement inhibition or modulation may be associated with possibility of a decreased immune defense against pathogens. Endophthalmitis is a severe adverse event with potential catastrophic visual outcome for participants. The rate of endophthalmitis for avacincaptad pegol in this trial was zero. This rate was lower than the reported endophthalmitis rate for C3 inhibition,38 which was higher than expected from comparable trials. This difference may be potentially due to the sample size or underlying biology, and should be further validated in a larger confirmatory phase 3 trial.
Best corrected visual acuity (BCVA) is generally a poor indicator of functional vision in patients with GA, since the fovea may be spared until the later stages of the disease. Accordingly, the visual disability that these participants experience is often underestimated. Further, the slow rate of VA loss over time precludes BCVA as an adequate primary efficacy endpoint in clinical trials for GA. Regulatory agencies have indicated that preventing photoreceptor loss, as reflected by GA area reduction measured by FAF, can be considered an adequate primary efficacy endpoint for GA secondary to AMD, given the established link between the photoreceptor loss and vision.
The rate of reported CNV for participants who received avacincaptad pegol was lower than what has been reported in the literature for complement C3 inhibition.38 This difference may be potentially due to the sample size or the underlying biology. Recent reports indicated that complement C3a may play an important role in complement homeostasis in the body, modulating complement activation based on the level of complement activity. It has been demonstrated that C3-CR3 (complement receptor 3) signaling may regulate the microglia-photoreceptor interactions in an animal model and potentially play an important role in maintaining the homeostasis in the retinal microenvironment. Further confirmatory larger trials are required to better understand this phenomenon.
One of the important features of this study was its screening trial design.37 A screening clinical trial design has all the design characteristics of a phase 3 pivotal clinical trial that include randomization, adequate double-masking, inclusion of a sham-control arm and a prespecified robust statistical analysis plan. In a screening trial, if the prespecified primary efficacy endpoint achieves the strength of evidence meeting the statistical significance, as was the case with avacincaptad pegol 2 mg and 4 mg, then the trial could potentially serve as one of the two required pivotal clinical trials for regulatory approval.
Based on a FDA guidance, a best fit curve analyses that shows a reduction in the rate of photoreceptor loss, reflected by reduction in GA area growth rate between baseline and at least two subsequent time points with intervals of 6 months or more between them, that exceeds measurement uncertainty is considered clinically meaningful.51 Accordingly, the beneficial effects of avacincaptad pegol 2 mg or 4 mg to reduce GA growth rate in this trial would be considered clinically meaningful.
Data from several trials and epidemiological studies indicate that effect of GA on visual function is an important public health problem. GA secondary to AMD currently impacts approximately 1.5 million individuals in in the United States and there are approximately 160,000 new cases per year, an incidence that is slightly higher than the number of new cases for wet AMD. These numbers will continue to increase as the size of the aging population continues to grow. Further, the development or progression of macular atrophy over time is an important cause of vision loss in eyes with wet AMD. If a confirmatory phase 3 trial shows a benefit of avacincaptad pegol to reduce GA growth rate, this therapy may be a potential treatment option for individuals with GA.
In conclusion, this study demonstrated for the first time that complement C5 inhibition slowed the progression of GA over 12 months in eyes with AMD. Avacincaptad pegol 2 mg and 4 mg were generally well tolerated in this elderly patient population. Since both avacincaptad pegol 2 mg and 4 mg met the primary endpoint with a similar efficacy and avacincaptad pegol 2 mg is administered as a single intravitreal injection while avacincaptad pegol 4 mg is administered as two injections, the confirmatory phase 3 pivotal trial will compare avacincaptad pegol 2 mg with a sham-control group (GATHER2 Study).
AUTHORS/CONTRIBUTORS:
- Glenn J. Jaffe, MD
- Keith Westby, MBA
- Karl G. Csaky, MD, PhD
- John Randolph, MD
- Harvey Masonson, MD
- Kourous A. Rezaei, MD
- Show all authors
Published in the American Academy of Ophthalmology Journal
Open Access. Published: August 31, 2020
DOI: doi.org/10.1016/j.ophtha.2020.08.027

